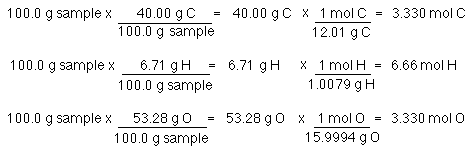SOLVED: What is the freezing point, in °C, of a solution made with 1.07 mol of CHCl₃ in 530.0 g of CCl₄ (Kf = 29.8 °C/m, normal freezing point, Tf = -22.9 °C)?
1) At 2257 K and 1.00 atm total pressure, water is 1.77 per cent dissociated at equilibrium by way of the reaction 2 H2O(g) <

Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are

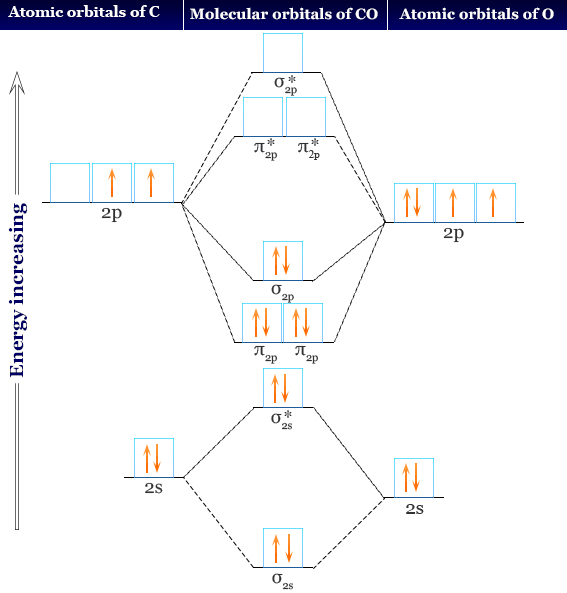
Draw the complete molecular orbital diagram for C^+_2 (form the molecular orbital diagram from the combination of a neutral C and a cationic C^+). | Homework.Study.com

SOLVED: How many of each the following contained in 100 grams of CO2 (m=44.01)? calculate mol of C,O and O2

Hydrogenation of fatty acids. a Oleic acid (0.5 mmol), SiNA-Pd (500 mol... | Download Scientific Diagram