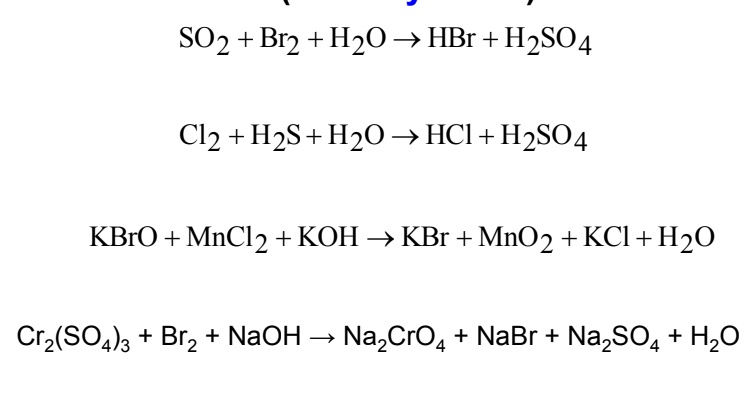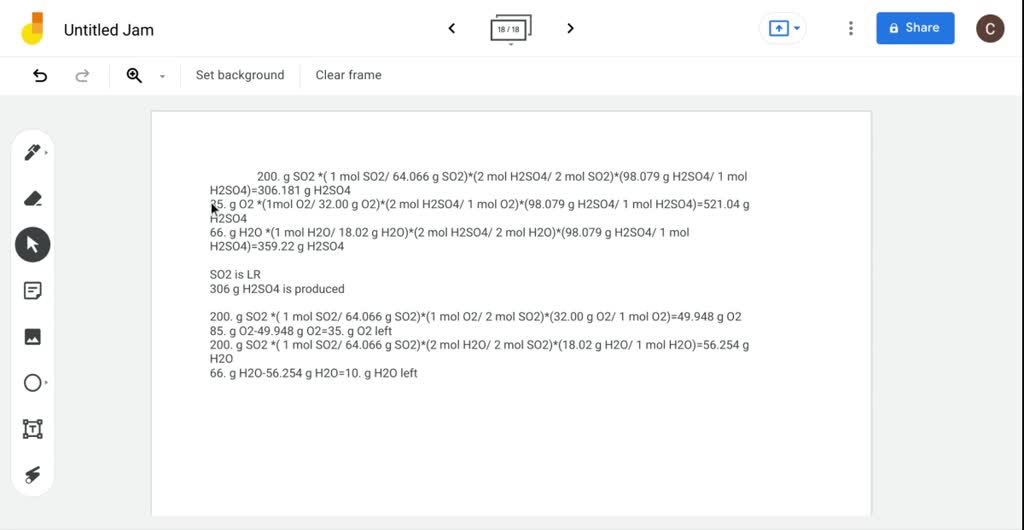
SOLVED: Sulfuric acid (H2SO4) forms in the chemical reaction2 SO2 +O2 + 2 H2O → 2 H2SO4 Suppose 400 g SO2, 175 g O2, and 125 g H2O are mixed and the

Volcanic sulfur dioxide (SO2) becomes sulfurous acid (H2SO3) in water... | Download Scientific Diagram
a) Complete the following reaction equations: (i) SO2 + MnO4- + H2O→ - Sarthaks eConnect | Largest Online Education Community
Stock Photos | Set Chemical Formula H2O, Pipette, Sulfur Dioxide SO2, Test Tube Flask On Stand, Helium, Mineral Ca Calcium And Icon. Vector
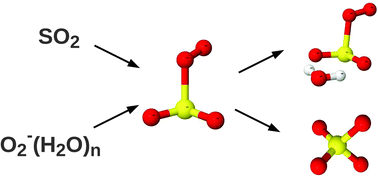
On the gas-phase reaction between SO2 and O2−(H2O)0–3 clusters – an ab initio study - Physical Chemistry Chemical Physics (RSC Publishing)

Balanced the following reactions:1. Cu + H2SO4→ CuSO4 + SO2↑ + H2O 2. Fe + H2O→Fe3O4 + H2↑ 3. CH4 + O2→ CO2+H2O