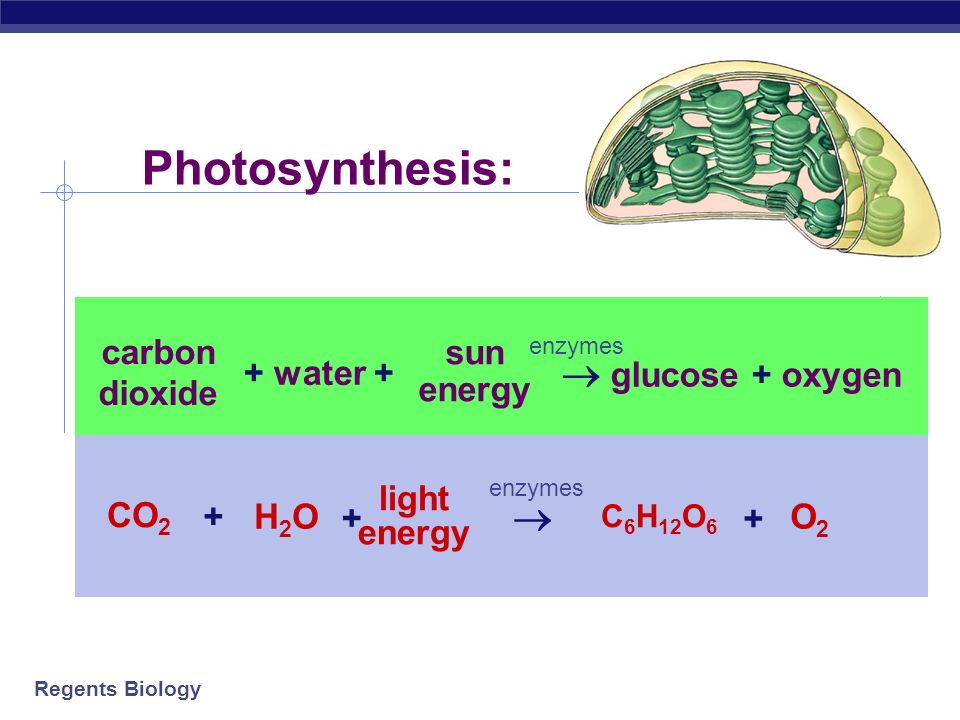
Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram

Performance and methane production characteristics of H2O–CO2 co-electrolysis in solid oxide electrolysis cells - ScienceDirect

Water (H2O) and carbon dioxide (CO2) both have one central atom with two atoms bonded to it. However, one is a polar molecule and one is not. Draw the Lewis structure for
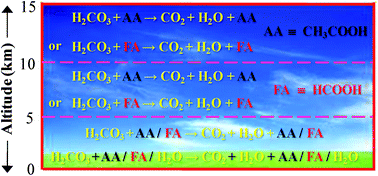
H2CO3 → CO2 + H2O decomposition in the presence of H2O, HCOOH, CH3COOH, H2SO4 and HO2 radical: instability of the gas-phase H2CO3 molecule in the troposphere and lower stratosphere - RSC Advances (

Write fully balanced equations for the following:(a) CO2 + H2O(b) Ca(OH)2+ CO2(c) SO2 + H2O(d) P205 + - Brainly.in
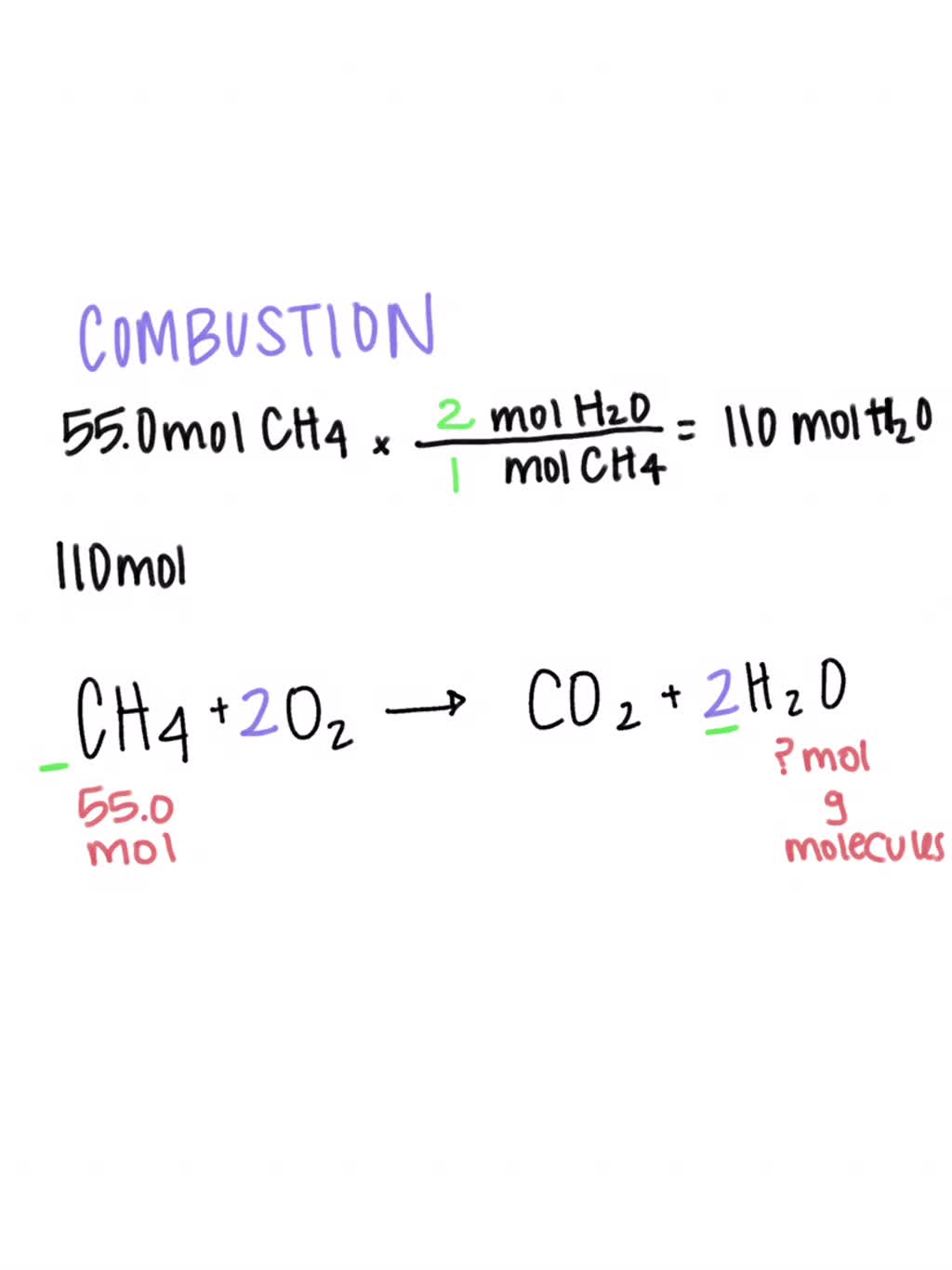
SOLVED: CH4 + O2 ——————- CO2 + H2O What type of reaction does this equation represent? Write and Balance the equation. Assume that there are 55.0moles of CH4. How many moles of