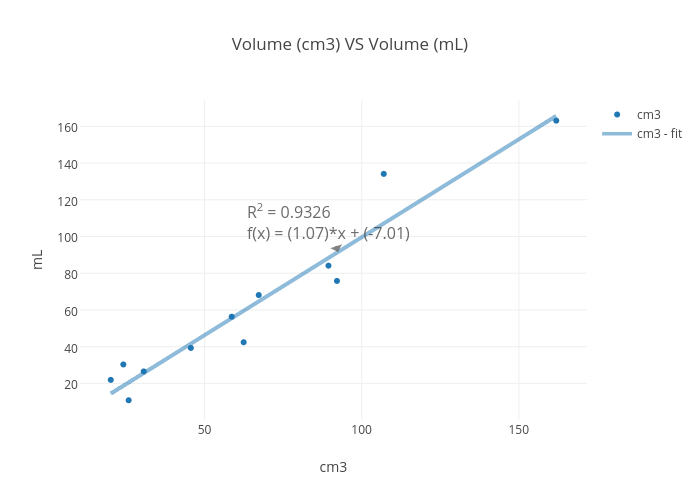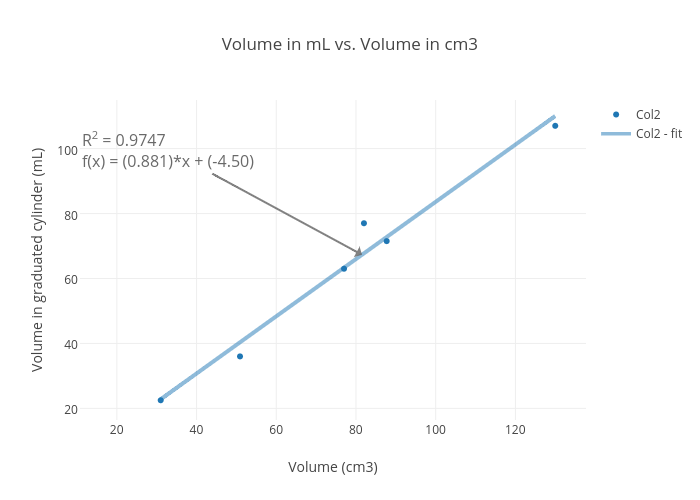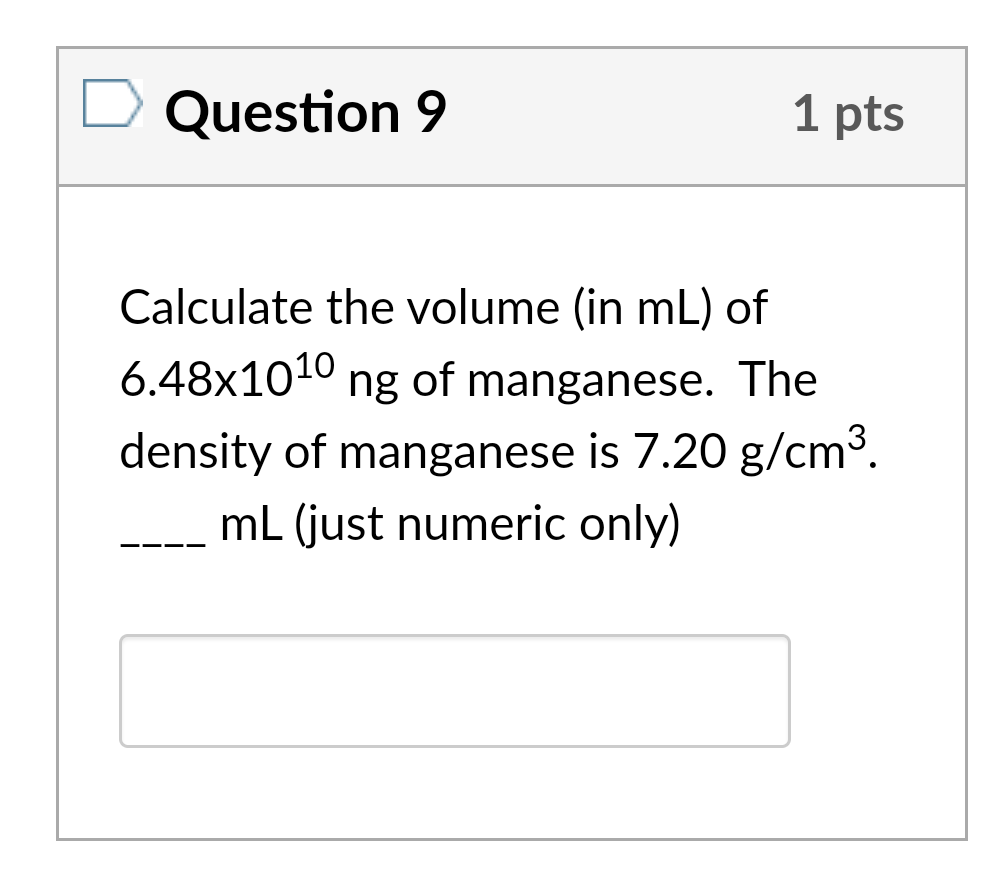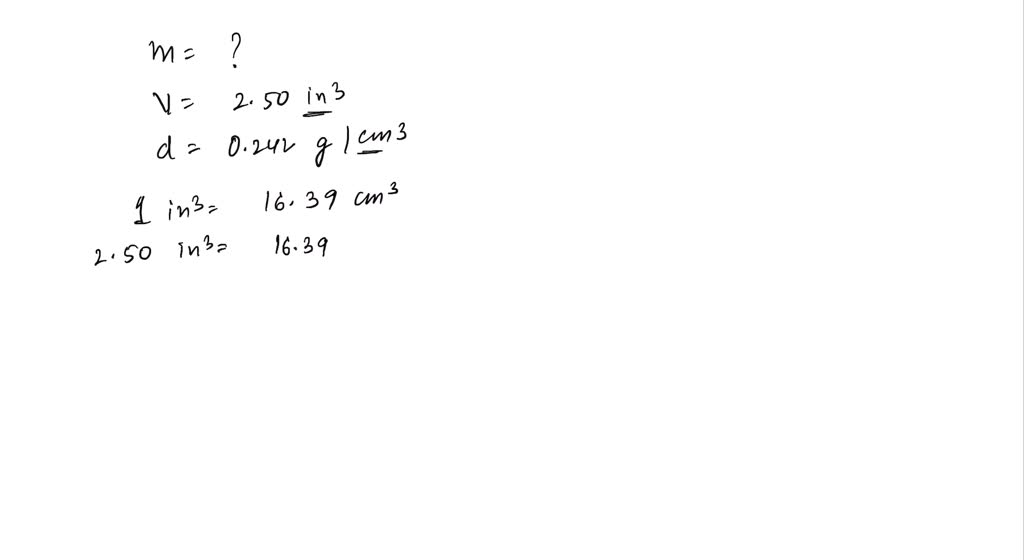
SOLVED: The mass unit associated with density is usually grams. If the volume (in mL or cm3) is multiplied by the density (g/mL or g/cm3) the volume units will cancel out, leaving
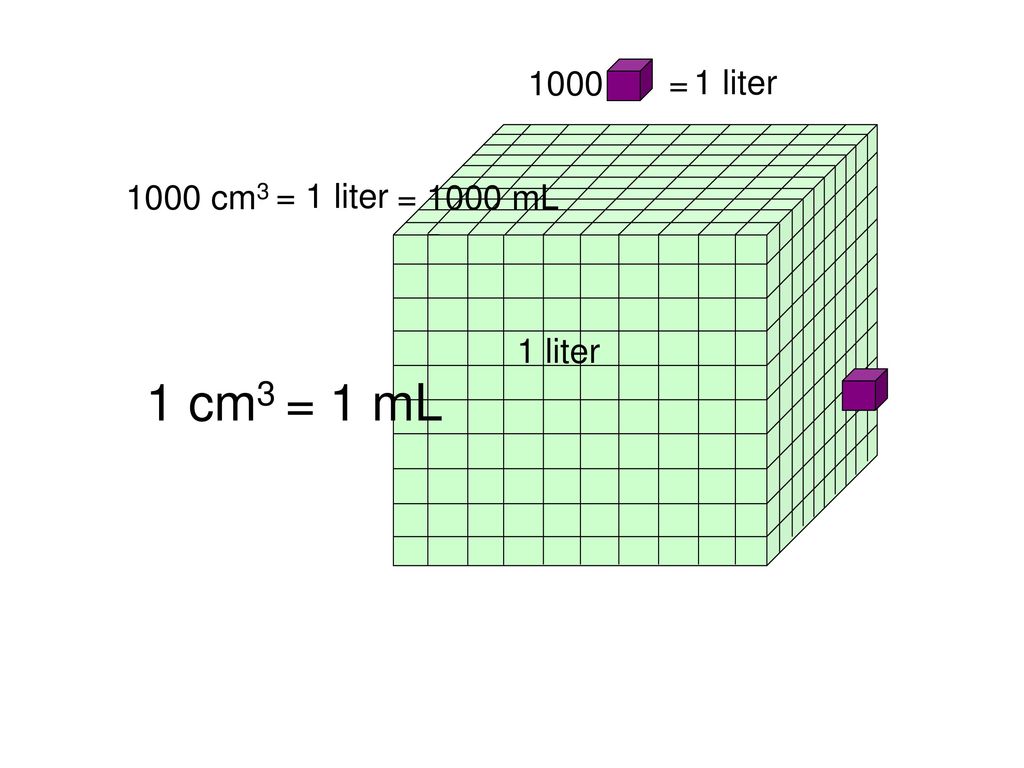
View question - Recall that 1 mL = 1 cm3. suppose that the volume of water in a gradated cylinder is 107.5 mL. A specimen of quartz, tied to a piece of stri